The exceptions found among the elements of group 2 (2A), group 15 (5A), and group 18 (8A) can be understood based on the electronic structure of these groups. As we go from left to right across a period, EAs tend to become more negative.
#ELECTRON AFFINITY CHART SERIES#
The second EA is the energy associated with adding an electron to an anion to form a 2– ion, and so on.Īs one might predict, it becomes easier to add an electron across a series of atoms as the effective nuclear charge of the atoms increases. Just as with ionization energy, subsequent EA values are associated with forming ions with more charge. However, for some elements, energy is required for the atom to become negatively charged, and the value of their EA is positive. Many of these elements have negative values of EA, which means that energy is released when the gaseous atom accepts an electron. This process can be either endothermic or exothermic, depending on the element.
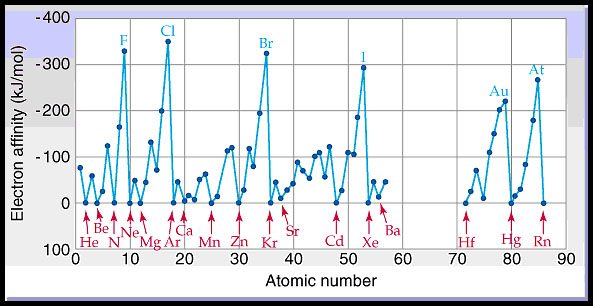
The electron affinity (EA) is the energy change for adding an electron to a gaseous atom to form an anion (negative ion).

This would increase the electron-electron repulsions and is therefore an energetically unfavourable process, which is also reflected in the less negative electron affinity compared to silicon. Unlike silicon, phosphorus has a half-filled p-subshell and the incoming electron needs to be paired with an electron already residing in the p-orbital. Interestingly, group 15 has less negative electron affinities than group 14. Thus, electron affinity values are either positive or less exothermic. The electron configuration indicates that the incoming electron needs to enter a higher-energy subshell. Thus, electron affinities for these elements are positive. The incoming electron has to be accommodated in the higher principal energy level, which is energetically unfavorable. In comparison, noble gases have a completely filled shell. Halogens have the most negative electron affinities, as the incoming electron helps to achieve noble gas configurations. Generally, moving across a period, electron affinities become more negative. This reduces the electron-electron repulsions, making it more attractive for an electron to be gained. In the chloride anion, however, the new electron is added into the third shell, occupying more space. But why?įluorine is the smallest atom of the halogens and an incoming electron experiences a significant repulsion from the electrons already present. In halogens, chlorine has a more negative electron affinity value than fluorine. Incoming electrons, therefore, experience less nuclear attraction leading to less negative electron affinities. Moving down group 1, the atomic size increases as the electrons occupy higher principal quantum numbers. Electron affinities, similar to ionization energies, show trends in the periodic table. In general, the greater the attraction between an atom and an added electron, the more negative the electron affinity. The negative sign indicates that it is an exothermic change.Īrgon, however, has a positive electron affinity, indicating that formation of an argon anion requires energy to be supplied. Electron affinity measures the ease of gaining an electron by an atom.įor example, the electron affinity of chlorine is −348.6 kJ/mol. When an electron is added to a gaseous atom, a change in energy is observed called electron affinity.
